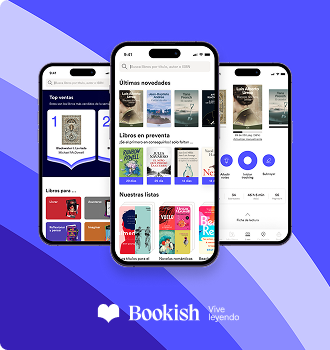
Detalls del llibre
Patient self-reported data has become increasingly important in today's clinical trials. Regulators and the industry have become increasingly aware of the limitations of recording patient reported outcomes data on paper. This book also reviews the FDA guidance, and provides a very contemporary view on this important subject.
Llegir més - Autor/a Brian Tiplady
- ISBN13 9780566087714
- ISBN10 0566087715
- Pàgines 296
- Any Edició 2010
- Fecha de publicación 28/10/2010
- Idioma Alemany, Francès
Ressenyes i valoracions
EPro: Electronic Solutions for Patient-Reported Data (Alemany, Francès)
- De
- Brian Tiplady
- 9780566087714
 4.7
4.7

















 Gràcies per comprar a llibreries reals!
Gràcies per comprar a llibreries reals!